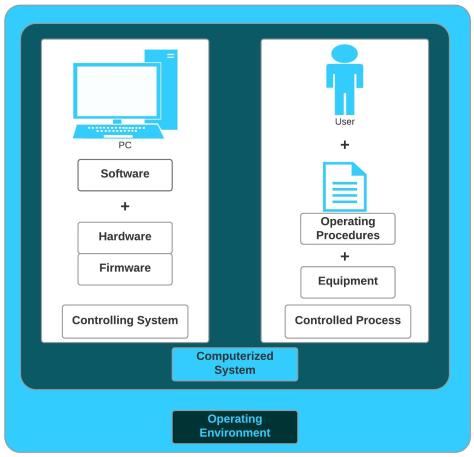
By definition, a computerized system consists of hardware, software, and network components and a controlled function or process that the computer system supports within a GxP operating environment as displayed in Figure 1. In other words, a computerized system is a computer system and a controlled process or function. The combination of the computer system and the controlled process is what produced the definition of a GxP computerized system. Among pharmaceutical industry professionals, especially validation engineers, the term is often shortened to simply ‘the system.’ Regardless, many computerized systems are subject to GxP regulations in the pharmaceutical industry. Since computerized systems help automate complex processes. Complex processes like those used in the manufacturing of drugs and biologics.
Figure 1: A GxP Computerized System
In a GxP environment, computerized systems cover a broad spectrum with examples including clinical trials data management, manufacturing resource planning, laboratory information management, process control network, manufacturing execution, document management, or automated laboratory equipment.
Note, the spelling may differ depending on which industry guidance you are looking at, computerized or computerised. Regardless of the spelling, the definition is the same.
What is the Controlling System?
The controlling system is all the computer-related elements of a computerized system. The terms controlling system and computer system are the same. The computer system consists of several parts; software, hardware, and firmware. Together, the software, hardware, and firmware make up the controlling system of a computerized system.
What is a Controlled Process?
A controlled process consists of procedures, people, and equipment; the second half of a computerized system. A controlled process is a workflow a computerized system supports within its GxP operating environment.
Controlled process is interchangeable with controlled function. The terms both mean a workflow associated with the computerized system.
In a pharmaceutical company’s production area, a controlled process could consist of a glasswasher as the equipment, manufacturing operators as the people, and the procedures for using the glasswasher.
Another example of a controlled process would be a laboratory instrument (equipment), a quality control test method (procedure), and the quality control analysts (users).
In either case, the controlled process represents a critical half of understanding and managing computerized systems. The controlled process should certainly not be overlooked during validation or operations if a manufacturer wants to be GxP compliant.
Computerized System = Computer System + Controlled Process
What is a Computer System?
Within the pharmaceutical industry, a computer system is all the computer hardware, firmware, installed devices, and software controlling the operation of the computer. Meaning the computer system makes up one-half of the computerized system. The computer system is analogous to the controlling system. Computer systems and computerized systems support a variety of GxP environments from clinical and commercial to research and development.
What is an GxP Operating Environment?
GxP operating environment is a fancy way of saying where the process is executed, or a system is used. The setting may be a lab, a clinic, or a manufacturing area that likely includes other computerized systems, equipment, systems, processes, and procedures. The term GxP comes into play when describing an operating environment since it covers multiple scenarios. GxP is an all-encompassing term for Good Clinical Practice (GCP), Good Laboratory Practice (GLP), Good Distribution Practice (GDP), Good Quality Practice (GQP), Good Manufacturing Practice (GMP), and Good Pharmacovigilance Practice (GPP).
How to define a computerized system?
Recall that the definition of a computerized system is a controlling system and a controlled process in the pharmaceutical industry. As a result, defining a particular computerized system involves first identifying the computer system and all of the elements of the controlled process. Meaning, all of the equipment, users, and procedures that the GxP computer system supports. Defining the controlled process helps establish the boundaries for the computerized system.
Which computerized systems are subject to GxP regulations?
Determining whether a computerized system is subject to GxP regulations depends on the system’s intended use. Intended use is a phrase that describes ‘how a system/product/process is used?’ and the environment in which it is used such as a research lab, a clinical setting, or commercial manufacturing. Depending on the intended use of the computerized system, the system may be subject to certain GxP regulations. GxP regulations like 21 CFR Part 11 for electronic records and electronic signatures or 21 CFR 211.68 to exercise appropriate controls related to records changes for commercial drug production.
Within the pharmaceutical industry, this type of assessment is commonly referred to as a GxP Determination. A GxP Determination is an assessment to decide whether the computerized system is GxP regulated and, if so, the specific regulations that apply. A GxP Determination is essentially asking several questions about the intended use of the system and the business processes the system will support. Questions like what functional areas will the computerized system support? Will the computerized system research and development, manufacturing, or, perhaps, a clinical setting? Answers to such questions will help determine applicable GxP regulations.
In the United States, the Food and Drug Administration (FDA) regulates drugs, biologics, and medical among several other products in accordance with the Code of Federal Regulations (CFR). For computerized systems, several relevant FDA regulations include the following:
- GxP Regulations for Computerized Systems
- Current Good Manufacturing Practices (CGMP) Regulations for Computerized Systems
- Pharmaceutical Drug
- Biological Product
- Medical Device Regulations
21 CFR Part 11 outlines the expectations for the use of electronic records and electronic signatures. As a result, Part 11 applies to such computer systems, meaning the regulations also apply to computerized systems. Next, the CGMP regulations above apply to the equipment used to manufacture drugs, biologics, and medical devices. Such equipment may be part of a defined computerized system and therefore applicable to the system.
Moreover, a GxP computerized system used in pharmaceutical drug manufacturing for Europe likely includes the EudraLex Volume 4 regulations from the European Commission. Within the EudraLex v4, the following sections are directly relevant to CGMP computerized systems:
Note, other regulations may apply. Therefore, be sure to consider the product (e.g., drug, biologic, medical device, or combination product), the relevant regulations (i.e., FDA, EMA, MHRA, etc.), the intended use of the computerized system as well as the data and record(s) generated from the system (e.g. production records, laboratory records, etc).
Related topics include….
Interested in courses on Computerized System Validation? Check out our training course here.
Need advice on your validation program or project? Click here for our consulting services.
References
- PIC/S Guidance on Good Practices for Computerized Systems in Regulated “GxP” Environments (PI 011-3) September 2007 (available at http://www.picscheme.org).
- ISPE GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems, International Society for Pharmaceutical Engineering (ISPE), Fifth Edition, February 2008, www.ispe.org
- US Code of Federal Regulations, Title 21, Food and Drugs
- 21 CFR Part 11 – Electronic Records, Electronic Signatures
- 21 CFR Part 211 – Current Good Manufacturing Practice for Finished Pharmaceuticals
- 21 CFR Part 606 – Current Good Manufacturing Practice for Blood and Blood Components
- 21 CFR Part 820 – Quality System Regulation (Medical Device)
- EUDRALEX Volume 4 – EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use. https://ec.europa.eu/health/documents/eudralex/vol-4_en